For technology focussed companies like in the medtech sector, developing new products and bringing them to market means being sustainable and competitive. The faster, more effective and productive the research and development teams work, the better it is for the innovative power of the company. Knowledge management supports not only the research and development department, but also the complete product life cycle through medical and regulatory affairs to customer service.
Promote product development with knowledge management
Knowledge management runs like a golden thread through all phases of innovation process and product development. To generate ideas for new products or improvements, all staff members need to be up-to-date with recent technological developments and trends of the market. This is already a fundamental challenge as current manual methods for extracting and synthesizing scientific, technical, and medical information do not scale to meet the demands imposed by the exponential growth of the unstructured evidence base. To date, there are more than 40,000 active scholarly peer-reviewed journals. Together, they publish more than 3 million articles per year, and the trend is rising.1
Apart from gathering information from external sources, it is important to maintain an active knowledge exchange between employees, so the company can benefit from each colleague’s strength. Especially during concept phase and development, sharing best practices, learning from past projects and the know-how of experts is essential for effective working procedures. But this only works when the knowledge is not locked up in the heads of people.
The need for managing knowledge has already been recognized in 2015 by the new version of ISO 9001. In the meantime the standardization experts have given this topic its own management standard, the ISO 30401 ‘knowledge management systems’. Within the strategic management measures, the knowledge management toolbox offers a wide range of methods and tools to identify, create and maintain knowledge and how to acquire it and make it available.
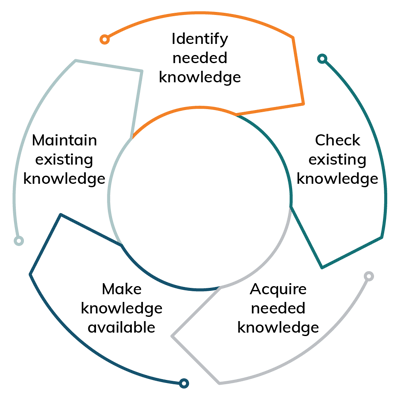
Adapted from: North, K. Wissensorientierte Unternehmensführung, Wissensmanagement im digitalen Wandel.
243–316 (2021) doi:10.1007/978-3-658-32771-2_8
Clinical evaluation as a central knowledge management process for medical device manufacturers
The Clinical evaluation is a systematic process that is mandatory for the medical device manufacturer for each medical device. The documentation of the clinical evaluation in a report is often carried out on behalf of the manufacturer by a contract research organization (CRO) or freelance medical writers. The structured and ongoing procedure consists of five essential steps which include, scoping and planning, collecting, appraising, and analyzing clinical data pertaining to a medical device. The purpose of the evaluation is an objective documentation that the device is safe and effective and in compliance with the general performance and safety requirements (GPSRs) according to the EU medical device regulation (MDR).
Within the Clinical Evaluation, the following sections focus on the clinical data (according to MDR and MEDDEV 2.7/1 rev 4):
- Clinical background, current knowledge, state-of-the-art
- Device and equivalent device specific clinical data (e.g. from scientific literature, unpublished manufacturer data)
- Analysis of the clinical data (e.g. safety, risk, benefit, performance)
During the initial or renewal of the device market approval, the clinical evaluation report is reviewed as part of the technical documentation by a Notified Body within the conformity assessment to obtain the CE-mark.
However, the Clinical Evaluation not only represents one of the most important regulatory documents, but also supports a central knowledge management process through the proactive, systematic and continuous process in collaboration with the various departments. It is also important to note that Clinical Evaluation is closely linked to other regulatory processes, such as risk management, post-marketing surveillance (PMS) and post-marketing clinical follow-up (PMCF).
Competitiveness through "Product Intelligence"
Therefore, the CER should not be regarded only as a regulatory requirement but as a device specific information base. In an ideal scenario, various stakeholders within the company, including R&D, product management, clinical affairs and sales keep the information up to date and available across functional boundaries within the organization. This information together with the experience of the employees represents the knowledge for a specific device within the company. As a result, this growing product intelligence together with the companies’ unique competencies and right acting will create a competitive advantage over time.
In general, gaining product intelligence should be a strong motivation for knowledge intensive industries, like medtech, in-vitro diagnostic, biotech and pharma. A strategic knowledge management approach together with AI-driven methods will be a core competency in during the era of exponential growth of data and information overload.
1 Johnson R, Watkinson A, Mabe M. The STM Report: An overview of scientific and scholarly publishing (Fifth Edition). The Hague: International Association of Scientific, Technical and Medical Publishers; 2018.

Robert Radloff
MBA, Founder of Curedatis
www.curedatis.com
Studied technical biology at the University of Stuttgart (Germany) with a focus on molecular biology and bioinformatics. Robert has more than 8 years’ pharmaceutical and medical device industry experience as a senior clinical research associate, product manager and medical writer in various therapeutic areas. The last year, Robert has created a prototype software solution; Curedatis. Curedatis is a medical language processing and "knowledge as a service" startup. Its goal is to realize the existing potential of big data within scientific publication with the use of smart workflows together with machine learning and natural language processing (NLP).

Gesa Krauss
Freelance Consultant
www.finfoma.de
Linkedin
As a freelance consultant, I take care of the evaluation, new or restructuring and optimization of information and knowledge management and topics related to data and process management in small and medium-sized companies. In more than 14 years of working in companies in the legal, business and tax consulting industries, I have become familiar with various organizational structures and corporate cultures and know what matters when information and knowledge are the key to a company's success. I love working with and analyzing data, as well as preparing it according to customer requirements. I am motivated by working with people and developing and realizing projects together with them.
Robert Radloff | MBA, Founder of Curedatis
Robert Radloff (MBA), founder of Curedatis, studied technical biology at the University of Stuttgart (Germany) with a focus on molecular biology and bioinformatics. Robert has more than 8 years’ pharmaceutical and medical device industry experience as a senior clinical research associate, product manager and medical...


